FamiCord Cell & Gene Manufacturing
FamiCord Cell & Gene Manufacturing, part of the FamiCord group, was founded in 2002 to meet a growing need in the marketplace for stem cell banking. After 12 years of optimizing the stem cell isolation and collection process from cord blood and processing of hundreds of thousands of products across the network, in 2014 we added Contract Development and Manufacturing Organization to our capabilities at our main facility in Warsaw. This new service has since gained traction and we were quickly supplying Cell Therapies to academic, research and pharmaceutical customers globally.

2002 year of establishment
We have since gained experience in development, manufacturing, storage, transport of drug products destined for over 20 clinical trials from our facilities.
Our mission is to use our expertise in cell manipulation, storage and banking purposes to develop the necessary tools required to bring cell therapies to our client’s patients in an effective, safe and timely manner.
Facilities & Systems
As the largest stem cell bank in Europe, FamiCord operates a network of laboratories, with 15 laboratories located in Europe, 4 GMP laboratories, 2 GMP accredited for the manufacture of ATMP material and 16 capable of storage, processing raw material and logistics support we are uniquely positioned for assisting the European market. Across the network, we currently store over 1,000,000 samples, cooperate with over 1,500 hospitals and have treated over 7,000 patients with product manufactured from our facilities.
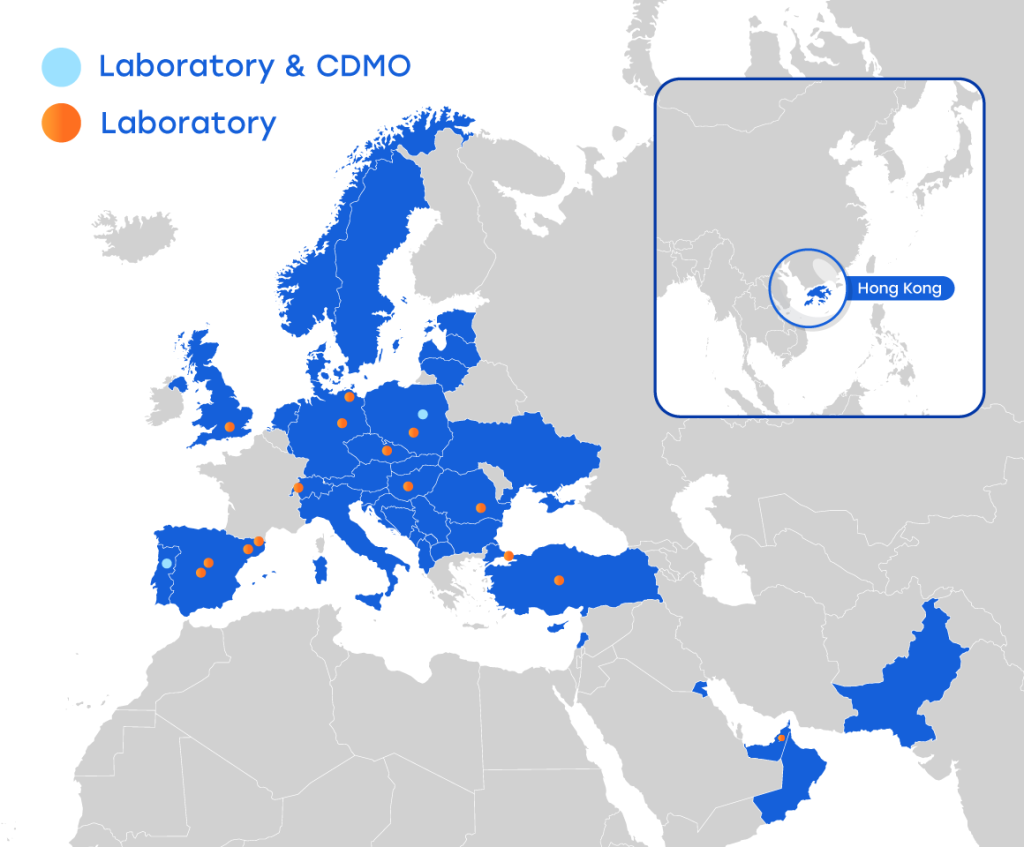
Our main facility is located in Warsaw, Poland. Our 1,100-square meter facility contains over 600 square meters of GMP production space. We have 16 separate production suites split into Tissue Engineered and Somatic Cell Therapy products, and Gene Modified Products. We offer single-batch autologous or multiple-batch allogeneic manufacturing for development purposes and clinical studies. We provide drug substance / product plus supporting documentation to streamline the IND submission process and carry you through the clinical trial process. The GMP suite design includes Class B clean rooms equipped with Class A cabinets. The facility is equipped to produce multiple ATIMP and ATMP-HE cell therapies from a variety of cell sources based on our clients’ specific requirements. Our Second GMP facility for GMP manufacturing is located in Cantanhede, Portugal. A 100m2 facility with 3 Class B suites for production, a Class C production suite and in-house analytical space for Cell Therapies. This is our Analytical Centre of Excellence with 20+ years of analyzing a wide range of cell therapies, with a particular focus on ATMP’s for clinical trials.
Our leadership team: helping to accellerate your cell therapy development
Callum King
Business Development Manager
Francisco dos Santos
DIRECTOR OF MANUFACTURING, PORTUGAL
Joanna Płaczkowska
DIRECTOR OF MANUFACTURING, POLAND
Karolina Zagórska
Global Director of quality assurance




